Analytical Solutions




Precision & Compliance
Analytical Experts
Quality Systems
Innovative Technologies
Pharmaceutical Analytical & Regulatory Solutions
Innosyna Pharma Lab Solution is an emerging pharmaceutical analytical service provider specializing in impurity profiling, working standards, and reference standards for instrument qualification including HPLC, GC, AAS, and ICP-OES.
We support pharmaceutical companies with high-precision analytical method development and validation for assay, dissolution, and related substances. Our expertise extends to chiral purity analysis, residual solvent testing, preservative analysis, and impurity isolation.
With strong domain knowledge in regulatory requirements, we assist in handling complex queries from global regulatory authorities during dossier submission and product market authorization, ensuring compliance and faster approvals.
Our Main Services
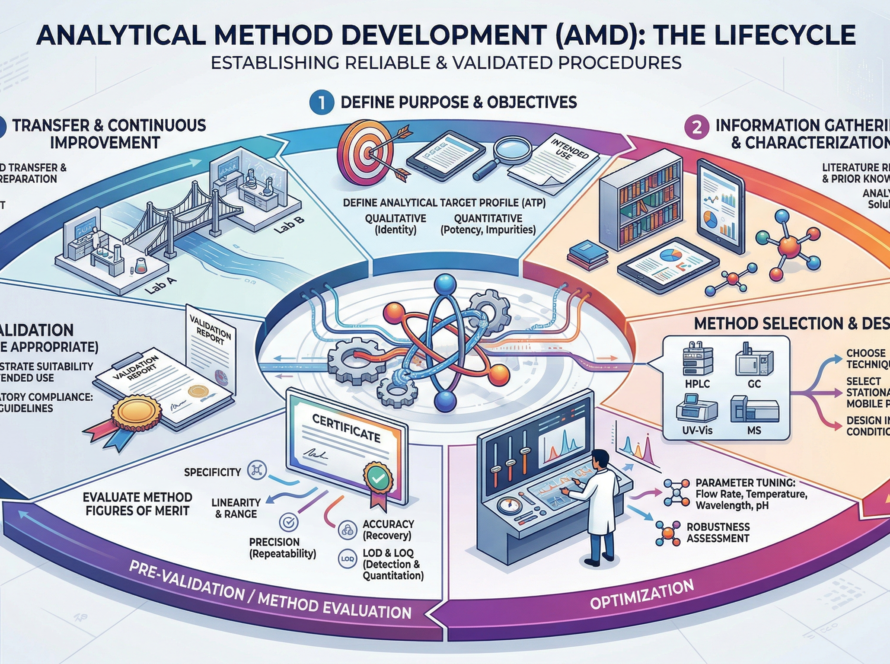
We provide end-to-end analytical solutions for pharmaceutical development, including impurity profiling, method development and validation, and advanced testing services using HPLC, GC, AAS, and ICP-OES technologies. Our solutions are designed to meet global regulatory standards and ensure reliable, high-quality results.
Analytical Method Validation (AMV) We deliver robust, ICH-compliant analytical method validation services ensuring accuracy, precision, reproducibility, and regulatory acceptance for pharmaceutical products. Our Capabilities Assay Method Validation (HPLC / GC)…
Reference Standards for Instrument Qualification We provide high-quality reference standards to ensure accurate calibration, qualification, and performance verification of analytical instruments. HPLC Precision standards for system suitability and calibration. GC…
Impurities, Intermediates & Working Standards We provide high-purity impurities, intermediates, and working standards to support pharmaceutical development, analytical testing, and regulatory compliance. Impurities Qualified impurity standards for identification, quantification, and…
Comprehensive evaluation of potential contaminants migrating from packaging materials and manufacturing systems to ensure product safety, quality, and regulatory compliance.
Accurate determination and validation of preservative content in pharmaceutical formulations
to ensure product stability, safety, and regulatory compliance.
Residual Solvents Analysis & AMV Accurate identification, quantification, and validation of residual solvents in pharmaceutical products to ensure safety, quality, and regulatory compliance as per global standards. Our Services Residual…
Our Experts
Dicta sunt explicabo. Nemo enim ipsam voluptatem quia voluptas sit aspernaturaut odit aut fugit, sed quia consequuntur. Dicta sunt explicabo. Nemo enim ipsam voluptatem quia voluptas.